|
Use the molar mass of glucose (to one decimal place, 180. Solid potassium chlorate, KClO 3, decomposes to form solid potassium chloride and diatomic oxygen gas. This is a requirement the equation must satisfy to be consistent with the law of conservation of matter. Write a balanced equation describing each of the following chemical reactions. 
Inspection shows that it is balanced as written, so the strategy outlined above can be adapted as follows:ġ. The chemical equation described in section 4.1 is balanced, meaning that equal numbers of atoms for each element involved in the reaction are represented on the reactant and product sides. A balanced equation is a chemical equation in which mass is conserved and there are equal numbers of atoms of each element on both sides of the equation. The initial step in solving a problem of this type is to write the balanced chemical equation for the reaction. If a typical 2 oz candy bar contains the equivalent of 45.3 g of glucose and the glucose is completely converted to carbon dioxide during the exam, how many grams of carbon dioxide will you produce and exhale into the exam room? Enter an equation of a redox chemical reaction and press the Balance button. You therefore decide to eat a candy bar to make sure that your brain does not run out of energy during the exam (even though there is no direct evidence that consumption of candy bars improves performance on chemistry exams). So, another one and another one right over there. Chemical reactions can be represented on paper with the help of chemical equations, an example for which is represented below (for the reaction between hydrogen gas and oxygen gas to form water). So this kind of equation is used for solving complicated equations. It is evident from the above example that the reactants and products of an equation are balanced. The solution to this would be: CH 4 + 2O 2 CO 2 + 2H 2 O. In order to balance it, were going to have six oxygens on the reactant side. Chemical equations were first formulated by the French chemist Jean Beguin in the year 1615. A question is given to balance the chemical equation: CH 4 + O 2 CO 2 + H 2 O. 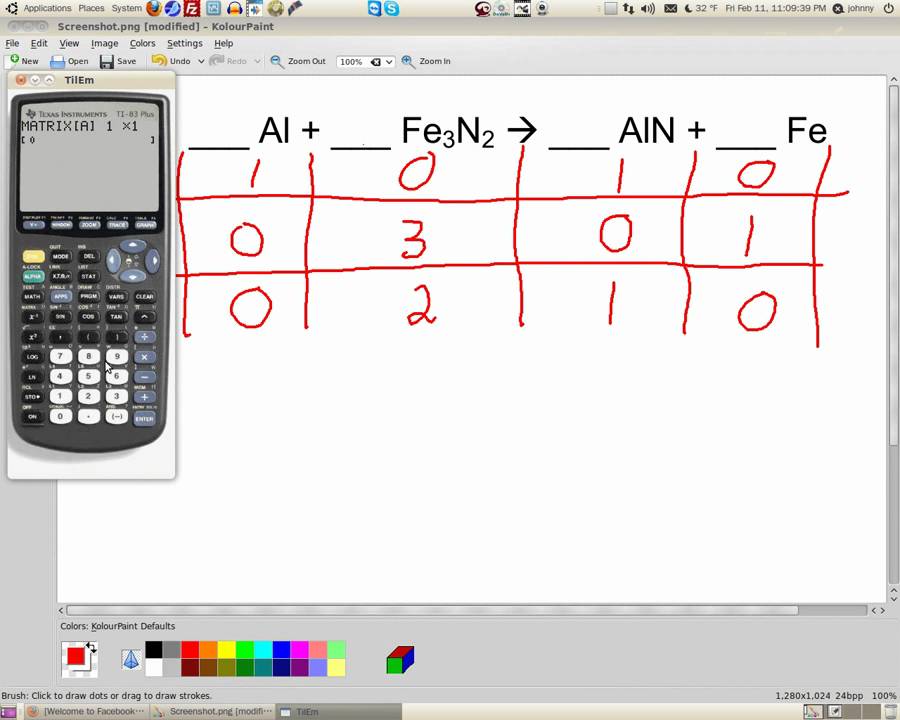 
Just before a chemistry exam, suppose a friend reminds you that glucose is the major fuel used by the human brain. On this side we have one, two, three, four, five, six oxygens.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
